News
EQS-HV: Merck KGaA: Korrektur: Bekanntmachung der Einberufung zur Hauptversammlung am 28.04.2023 in Darmstadt mit dem Ziel der europaweiten Verbreitung gemäß §121 AktG
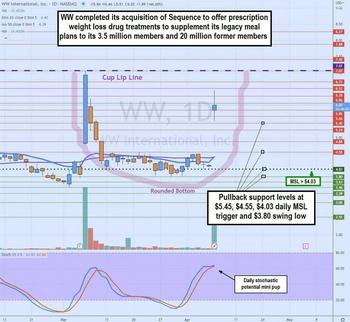
Weight Watchers Reshapes Itself With A Game-Changer Acquisition
Key Points
- Weight Watchers closed its $132 million acquisition of telehealth weight loss platform Sequence Inc. sending shares higher by over 25% on over 37 million shares volume.
- Sequence
EQS-HV: Merck KGaA: Bekanntmachung der Einberufung zur Hauptversammlung am 28.04.2023 in Darmstadt mit dem Ziel der europaweiten Verbreitung gemäß §121 AktG

EQS-HV: Bayer Aktiengesellschaft: Bekanntmachung der Einberufung zur Hauptversammlung am 28.04.2023 in Virtuell/Leverkusen mit dem Ziel der europaweiten Verbreitung gemäß §121 AktG
EQS-Adhoc: Bayer Aktiengesellschaft: William N. Anderson folgt auf Werner Baumann als Vorstandsvorsitzender der Bayer AG
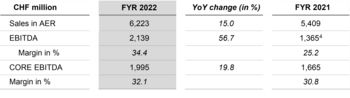
Lonza Delivers Strong 2022 Performance with 15.1% CER Sales Growth and 32.1% CORE EBITDA Margin

Lonza erzielt 2022 ein starkes Ergebnis mit 15.1% Umsatzwachstum (kWk) und 32.1% Kern-EBITDA-Marge

Lonza Appoints Daniel Palmacci as President of the Cell & Gene Division

Lonza erzielt im ersten Halbjahr 2022 ein solides Ergebnis mit einem Umsatzwachstum von 16.8% (kWk) und einer Kern-EBITDA-Marge von 33.1%
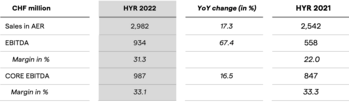
Lonza Delivers Solid Performance in H1 2022 with 16.8% CER Sales Growth and 33.1% CORE EBITDA Margin

Maria Soler Nunez appointed as Head, Group Operations
Hardman & Co Life Sciences Research: Long-term pharmaceutical industry cost and EBIT analysis
Hardman & Co Life Sciences Research: Long-term cost and EBIT analysis of the pharmaceutical industry

Johnson & Johnson Update: ist die Abspaltung gesund oder drohen Probleme?

Abbvie Inc - Technical Analysis

Pfizer Invites Public to View and Listen to Webcast of Pfizer Discussion at Healthcare Conference
Pfizer Inc. (NYSE: PFE) invites investors and the general public to view and listen to a webcast of a discussion with Bob Smith, Vice President, Pfizer Rare Disease, at the 4th Annual Evercore ISI

Follow-Up Data From Phase 3 Trial of Pfizer-BioNTech COVID-19 Vaccine Support Safety and High Efficacy in Adolescents 12 Through 15 Years of Age
Pfizer Inc. (NYSE:PFE) and BioNTech SE (Nasdaq:BNTX) today announced topline results from a longer-term analysis of the safety and efficacy of their COVID-19 vaccine in individuals 12 through 15

Pfizer and BioNTech Receive Expanded U.S. FDA Emergency Use Authorization of COVID-19 Vaccine Booster to Include Individuals 18 and Older
Pfizer Inc. (NYSE: PFE) and BioNTech SE (Nasdaq: BNTX) today announced that the U.S. Food and Drug Administration (FDA) has expanded the emergency use authorization (EUA) of a booster dose of the

Pfizer’s XELJANZ® (tofacitinib) Receives Marketing Authorization in the European Union for the Treatment of Active Ankylosing Spondylitis
Pfizer Inc. (NYSE: PFE) announced today that the European Commission has approved XELJANZ® (tofacitinib) 5 mg twice daily for the treatment of adults with active ankylosing spondylitis (AS) who

Pfizer to Provide U.S. Government with 10 Million Treatment Courses of Investigational Oral Antiviral Candidate to Help Combat COVID-19
Pfizer Inc. (NYSE: PFE) today announced an agreement with the U.S. government to supply 10 million treatment courses of its investigational COVID-19 oral antiviral candidate, PAXLOVID™

Pfizer Completes Acquisition of Trillium Therapeutics
Pfizer Inc. (NYSE: PFE) today announced the successful completion of its acquisition of Trillium Therapeutics, a clinical stage immuno-oncology company developing innovative therapies for the

Pfizer Announces Retirement of Chief Financial Officer Frank D’Amelio
Pfizer Inc. (NYSE: PFE) today announced that Frank D’Amelio will retire from his position as Chief Financial Officer and Executive Vice President of Global Supply at Pfizer after a nearly 15 year

Pfizer Seeks Emergency Use Authorization for Novel COVID-19 Oral Antiviral Candidate
Pfizer Inc. today announced it is seeking Emergency Use Authorization (EUA) of its investigational oral antiviral candidate, PAXLOVID™ (PF-07321332; ritonavir), for the treatment of mild to

Pfizer and The Medicines Patent Pool (MPP) Sign Licensing Agreement for COVID-19 Oral Antiviral Treatment Candidate to Expand Access in Low- and Middle-Income Countries
Pfizer Inc. (NYSE: PFE) and the Medicines Patent Pool (MPP), a United Nations-backed public health organization working to increase access to life-saving medicines for low- and middle-income








.png?locale=us)

