News

NANOBIOTIX Announces 18.1 Month Median Overall Survival for 41 Evaluable Elderly and Frail Patients With HNSCC in Phase I Expansion Evaluating Nbtxr3 as a Single Agent Activated by Radiotherapy
Regulatory News:
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20211024005038/en/
Figure 1: Best Observed Target Lesion Response by

BOIRON : Activity in the third quarter of 2021
(Unaudited data)
Development of business in the third quarter (variation at current exchange rates)
In thousands of euros 1st quarter 2nd quarter 3rd quarter 2021 2020 Var.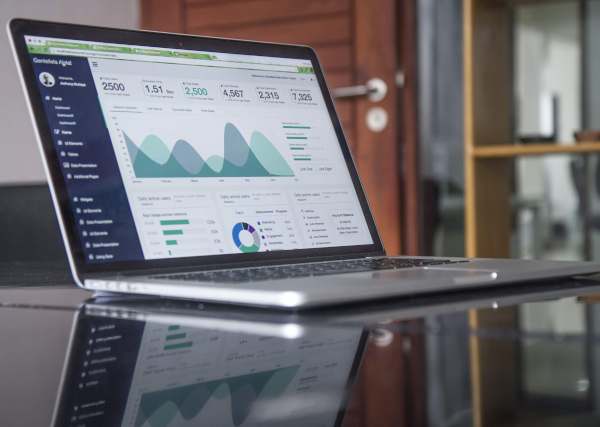
Valneva Aktie - Impfstoff ohne Nebenwirkungen?

NANOBIOTIX Provides Third Quarter Operational and Financial Update
Regulatory News:
NANOBIOTIX (Euronext: NANO - NASDAQ: NBTX - the ‘‘Company’’), a late-clinical stage biotechnology company pioneering physics-based approaches to expand treatment possibilities

Poxel Provides Corporate Update and Reports Cash and Revenue for the Third Quarter and Nine Months 2021
POXEL SA (Euronext – POXEL - FR0012432516), a clinical stage biopharmaceutical company developing innovative treatments for chronic diseases with metabolic pathophysiology, including non-alcoholic
Ipsen erweitert seine präklinische Forschungs- und Entwicklungspipeline für Onkologie im Rahmen einer exklusiven weltweiten Zusammenarbeit mit Accent Therapeutics um ein weiteres Programm mit Fokus auf das RNA-modifizierende Protein METTL3
Haftungsausschluss: Nur für internationale Medien und Investoren bestimmt
Ipsen (Euronext: IPN; ADR: IPSEY) und Accent Therapeutics (Accent) haben eine exklusive weltweite
Ipsen Adds Another Program Into Its Pre-Clinical R&D Oncology Pipeline Through an Exclusive Worldwide Collaboration With Accent Therapeutics, Targeting the RNA Modifying Protein, METTL3
Regulatory News:
Disclaimer: Intended for international media and investor audiences only
Ipsen (Euronext: IPN; ADR: IPSEY) and Accent Therapeutics (Accent) have signed an exclusive

GenSight Biologics Announces FDA Grant of Fast Track Designation for Optogenetic Therapy GS030 as Treatment for Retinitis Pigmentosa
Regulatory News:
GenSight Biologics (Euronext: SIGHT, ISIN: FR0013183985, PEA-PME eligible), a biopharma company focused on developing and commercializing innovative gene therapies for retinal

ABIONYX Pharma Initiates Discussions With IRIS Pharma, a World Leader in Preclinical and Clinical Ophthalmology Research, With a View to a Possible Strategic Deal
Regulatory News:
ABIONYX Pharma (Paris:ABNX) (FR0012616852 - ABNX - PEA PME eligible), a next-generation biotech company dedicated to the discovery and development of innovative therapies

Nanobiotix: Statement of Total Voting Rights and Shares Forming the Company’s Share Capital
Regulatory News:
Nanobiotix (Paris:NANO) (NASDAQ:NBTX):
Market: Euronext Paris
Compartment: B
ISIN code: FR0011341205
Website: www.nanobiotix.com
Date
Number of Shares
Outstanding

NANOBIOTIX to Present First Survival Data From Priority Head and Neck Cancer Program Among Five Presentations at the 2021 Annual Meeting of the American Society for Radiation Oncology
Regulatory News:
NANOBIOTIX (Euronext: NANO – NASDAQ: NBTX – the ‘‘Company’’), a late-stage clinical biotechnology company pioneering physics-based approaches to expand treatment possibilities

GenSight Biologics Announces the Publication of a Review of Gene Therapy Trials for LHON in International Ophthalmology Clinics
Regulatory News:
GenSight Biologics (Euronext: SIGHT, ISIN: FR0013183985, PEA-PME eligible), a biopharma company focused on developing and commercializing innovative gene therapies for retinal

Transgene and BioInvent to Present Preclinical Data on BT-001 Oncolytic Virus at SITC 2021
Regulatory News:
Transgene (Euronext Paris: TNG) (Paris:TNG), a biotech company that designs and develops virus-based immunotherapeutics against cancer, and BioInvent International AB

Poxel Announces its Participation at Investor Conferences in October 2021
Regulatory News:
POXEL SA (Euronext – POXEL – FR0012432516), a clinical stage biopharmaceutical company developing innovative treatments for chronic serious diseases with metabolic

NANOBIOTIX Announces Red Journal Publication of Preclinical Data Showing Radioenhancer NBTXR3 May “Reprogram” the Tumor Microenvironment to Overcome Anti-PD-1 Resistance and Evoke Abscopal Effect
Regulatory News:
NANOBIOTIX (Euronext : NANO –– NASDAQ: NBTX – the ‘‘Company’’), a late-clinical stage biotechnology company pioneering nanophysics-based approaches to expand treatment

Transgene’s Two Innovative Platforms Progressing Well - Financial Visibility Extended Until End 2023
Regulatory News:
Transgene (Paris:TNG) (Euronext Paris: TNG), a biotech company that designs and develops virus-based immunotherapies for the treatment of cancer, today publishes its financial

Transgene Presents Data From Phase I Clinical Trial Confirming the Potential of the Oncolytic Virus TG6002
Regulatory News:
Transgene (Paris:TNG) (Euronext Paris: TNG), a biotech company that designs and develops virus-based immunotherapeutics against cancer, today announces the presentation of data

GenSight Biologics Announces Positive Data Safety Monitoring Board Review of PIONEER Phase I/II Clinical Trial of GS030 as Optogenetic Treatment for Retinitis Pigmentosa
Regulatory News:
GenSight Biologics (Euronext: SIGHT, ISIN: FR0013183985, PEA-PME eligible), a biopharma company focused on developing and commercializing innovative gene therapies for retinal

Transgene Announces Upcoming Investor Meetings
Regulatory News:
Transgene (Euronext Paris: TNG), a biotech company that designs and develops virus-based immunotherapeutics against cancer, today announces that Management will participate in

Nanobiotix: Statement of Total Voting Rights and Shares Forming the Company’s Share Capital as of August 31, 2021
Regulatory News:
Nanobiotix (Paris:NANO) (NASDAQ:NBTX):
Market: Euronext Paris
Compartment: B
ISIN code: FR0011341205
Website: www.nanobiotix.com
Date
Number of Shares
Outstanding

BOIRON : 2021 Half-Year Results
(Data have been the subject to a limited review by the statutory auditors)
The Board of Directors, at its meeting of September 8, 2021, made up the half-year statutory and consolidated financial

GenSight Biologics Announces Publication Analyzing Visual Parameters of ND4-LHON Subjects Before LUMEVOQ® Treatment in Phase III Trials
Regulatory News:
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20210908006154/en/
Figure 1. BCVA in untreated ND4-LHON patients as a

NANOBIOTIX Provides Business Update and Reports Financial Results for the First Half of 2021
Regulatory News:
NANOBIOTIX (Euronext: NANO –– NASDAQ: NBTX – the ‘‘Company’’), a late-clinical stage biotechnology company pioneering physics-based approaches to expand treatment possibilities

Nanobiotix to Present at the H.C. Wainwright 23rd Annual Global Investment Conference
Regulatory News:
NANOBIOTIX (Euronext: NANO – NASDAQ: NBTX – the ‘‘Company’’), a late-clinical stage biotechnology company pioneering physics-based approaches to expand treatment possibilities

UK MHRA Grants GenSight Biologics’ LUMEVOQ® Ophthalmic Gene Therapy Promising Innovative Medicine Designation
GenSight Biologics (Paris:SIGHT) (Euronext: SIGHT, ISIN: FR0013183985, PEA-PME eligible), a biopharma company focused on developing and commercializing innovative gene therapies for retinal







